Genome & Company
Contact
Genome & Company
7F GWANGGYO FLAX DESIAN, 50 Changnyong-daero 256beon-gil, Yeongtong-gu, Suwon-si, Gyeonggi-do, Republic of Korea ,16229
Consent to collection and use of personal information (Required)
Genome & Company (the ”Company”) regards the protection of personal information of our users very important, and complies with relevant laws, including the Personal Information Protection Act and the Act on Expansion of Dissemination and Promotion of Utilization of Information System. The Company hereby informs the purpose and method of using the personal information provided by our users through the Company’s website, and what measures are taken to protect the personal information. If the Company revises any of this Private Policy, it will be notified through the notices on the Company’s website (or individually notified).
1. Items of Personal Information Collected and the Collection Method
The Company collects the following personal information to operate its Contact Service via the Company’s website (whereby the users can make general inquires, inquiry for business development, IR inquiry, media inquiry, etc.).
- Essential Information: name, e-mail address, phone number, organization, content of inquiry
- Collection method: The Company collects the personal information through the inquiries submitted via the Contact Bulletin Board on the Company’s website.
You have the right to refuse the ‘collection of essential information’ as well as the ‘collection of optional information’. If you refuse the collection of essential information, however, you may be restricted for using the Contact Service.
The following information may be automatically generated and collected in the process of using the website.
- IP Address, cookie, date and time of visit, records of service use
2. Purpose of Collecting Personal Information
The Company uses the collected personal information for the following purposes.
- Personal verification, personal identification, delivery of inquiries and notification of results (answers and responses), etc. related to the use of Contact Service
3. Processing of Personal Information and Retention Period
In principle, the collected personal information is destroyed without any delay after achieving the purpose of collecting and using personal information. However, the Company may keep users’ information for a certain period of time as required by relevant laws.
- Retained information: personal information submitted with the inquiry, content of the inquiry, and ensuing responses
- Relevant Law: Act on the Consumer Protection in Electronic Commerce, etc.
- Retention Period: three years (records related to resolution of consumer complaints or disputes)
4. Destruction of Personal Informatio
In principle, the information is destroyed without any delay after achieving the purpose of collecting and using personal information. The procedure and method of destruction are as follows.
1) Procedure
The personal information submitted is transferred to a separate database (in case of paper, separate cabinet) after achieving the purpose and kept for a certain period of time according to the internal policy and other relevant laws, before it is destroyed. The personal information transferred to separate database shall not be used any other purposes other than for keeping in accordance with laws.
2) Destruction Method
Personal information saved in the form of electronic file shall be deleted by using technical method that cannot reproduce the records. Personal information printed in the paper shall be destroyed by shredding with a shredder or incinerating it.
5. Consignment of Personal Information Processing
The Company does not consign personal information to a third party. If consignment occurs in the future, it shall be notified through website and the contents of this Privacy Policy shall be updated.
6. Rights of Users and Their Legal Representatives
Users and their legal representative (for the users below the age of 14) may make an inquiry related to or request deletion or modification of their own personal information. In this case, please contact the Company’s Data Protection Manager (see Section 7 of this Privacy Policy) in writing, by phone, or by e-mail so that we can take measures without any delay after verifying your identification.
In case users request the correction of any errors in personal information, we shall not use or provide the personal information until such correction is made. In addition, in case incorrect personal information is already provided to a third party, we will notify the third party the result of correction without delay.
The Company processes and destroys personal information at the request of users or their legal representatives in accordance with Section ‘3. Processing of Personal Information and Retention Period’ and Section ‘4. Destruction of Personal Information’, and such information shall not be opened or used for any other purposes.
7. Information on Data Protection Officer and Manager
The Company designates data protection officer and manager for managing personal information as follows to protect users’ personal information related to inquiries.
Data Protection Officer
Name: Seo Yeong-jin
Department: Business management
Position: Vice-president
Telephone: 031-620-0150
Email: youngjin.seo@genomecom.co.kr
Data Protection Manager
Name: Seol Su-gi
Department: PR team
Position: Team Lead
Telephone: 070-4821-3840
Email: sugi@genomecom.co.kr
You can submit to the personnel above any inquiries related to your personal information submitted to the Company while using our web service. The Company will quickly provide sufficient responses to your inquiries.
Please contact the below agencies if you need report or counseling for any infringement of personal data privacy.
- Personal Information Infringement Report Center (privacy.kisa.or.kr / 118 without area code)
- Personal Information Dispute Mediation Committee (kopico.go.kr / 1833-6972)
- Cyber Investigation Department, Supreme Prosecutor’s Office (www.spo.go.kr / 1301 without area code)
- Cyber Bureau of the National Police Agency (http://cyberbureau.police.go.kr / 182 without area code)
8. Notification
We will notify users any additions, deletions, and modification on the current Privacy Policy in advance through ‘Notice’ on the Company’s website.
9. Other Websites
We inform you that any collection of personal information by other websites which may be hyperlinked on the Company’s website is not the subject of this Privacy Policy.



Pipeline
GNOCLE™ Platform
Genome & Company has established a new drug development platform, GNOCLE™ and effectively conduct research and development in innovative microbiome and novel target based therapeutics. A such unique “Bed-to-Bench” approach using real-world clinical data collected from research institutions and hospitals significantly enhances the success probability of new drug development.
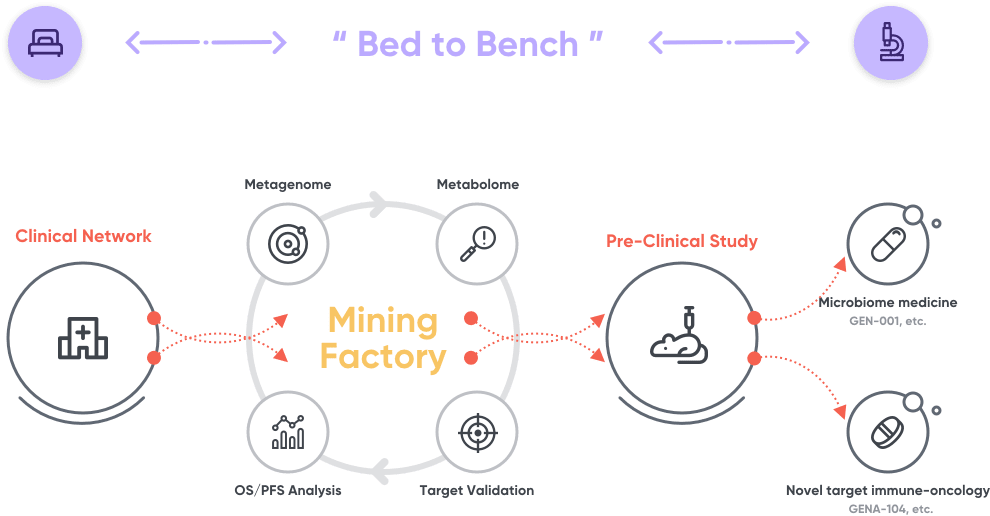
- 01 Establishment of clinical data base
- [Microbiome] Library of microbiome samples from patients with disease that has low responses to the existing medicines. Genome & Company owns over 10,000 clinical samples in cancer, anticancer rash, atopy, dementia, infertility, cognitive skills.
- [Novel target] Library of novel targets that are expressed from the refractory patients identified through the analysis of cancer tissues from the cancer patients treated by existing immunotherapy.
- 02 Discovery of bacterial strains for microbiome therapeutics and novel target verification
- [Microbiome] Identification of the mechanism of action through multi-omics analysis using real-world clinical samples and discovering bacterial strain candidates for microbiome therapeutics through in vitro/in vivo assays.
- [Novel target] Verification of novel targets by comparing prognosis of patients in accordance with the expression level of the target and identifying anti-cancer efficacy and mechanism of action (followed by new drug development as a new immunotherapy candidate)
[Pipeline] New biologics
*Click the pipeline name in the table to find more details
| Modality | Pipeline | Tartget | Indication | Developmental Status | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Target | Hit | Lead | Nonclinical candidate | IND enabling | Phase I | Partner | ||||
|
ADC
|
Debio 0633
|
N/D | Solid tumors | License-Out(May 2024) |
 |
|||||
|
GENA-104
|
CNTN4 | Solid tumors | ||||||||
| GENA-120 | ITGB4 | Solid tumors | ||||||||
| GENB-120 | ITGB4 TROP2 |
Solid tumors | ||||||||
| GENA-121 | N/D | Solid tumors | ||||||||
| ADC Programs | N/D | N/D | ||||||||
|
mAb(Immuno-oncology)
|
EP0089 | CNTN4 | Solid tumors |
KDDF Funded IND Approval (MFDS)
|
License-Out(Feb 2025) |
 |
||||
|
GENA-119
|
APP | Solid tumors |
KDDF Funded
|
|||||||
|
NCE
|
GENC-116 | NUAK1 | Fibrosis |
KDDF Funded
|
||||||
* N/D, not disclosed; mAb, monoclonal antibody; ADC, antibody-drug conjugate; Nce, new chmical entity
* For out-licensed pipeline assets, research and development is conducted by the respective partners; therefore, actual development progress may differ from the information presented herein.
GEN-001 Immune-oncology microbiome treatment

Genome & Company's lead pipeline GEN-001 is the first candidate for an immuno-oncology microbiome therapeutic in Korea.
GEN-001 is being developed as an orally administered microbiome therapeutic with Lactococcus lactis (L. lactis), a single strain bacteria isolated from a healthy human. Its anticancer efficacy was proven by activating immune activity of cancer cells. In pre-clinical setting, immune-anticancer effect was confirmed in both monotherapy and combination therapy with existing immunotherapy (immune checkpoint inhibitors).
Currently, GEN-001 is undergoing phase 2 clinical trials in combination with Merck KGaA's Bavencio® for gastric cancer. Additionally, it is also undergoing phase 2 clinical trials in combination with MSD's Keytruda® for cholangiocarcinoma.
- [Partnership]
-
- Clinical Trial Collaboration and Supply Agreement with Merck KGaA - Clinical study for combination therapy of 'GEN-001' and immune checkpoint inhibitor (BAVENCIO®) (Korea)
- Clinical Trial Collaboration and Supply Agreement with MSD - Clinical study for combination therapy of 'GEN-001' and immune checkpoint inhibitor (KEYTRUDA®) (Korea)
- [Commercialization]
- - License-out Agreement in East Asia with LG Chem, Ltd.
*Find more details in the Press Release section of the website.



SB-121Brain diseases microbiome treatment
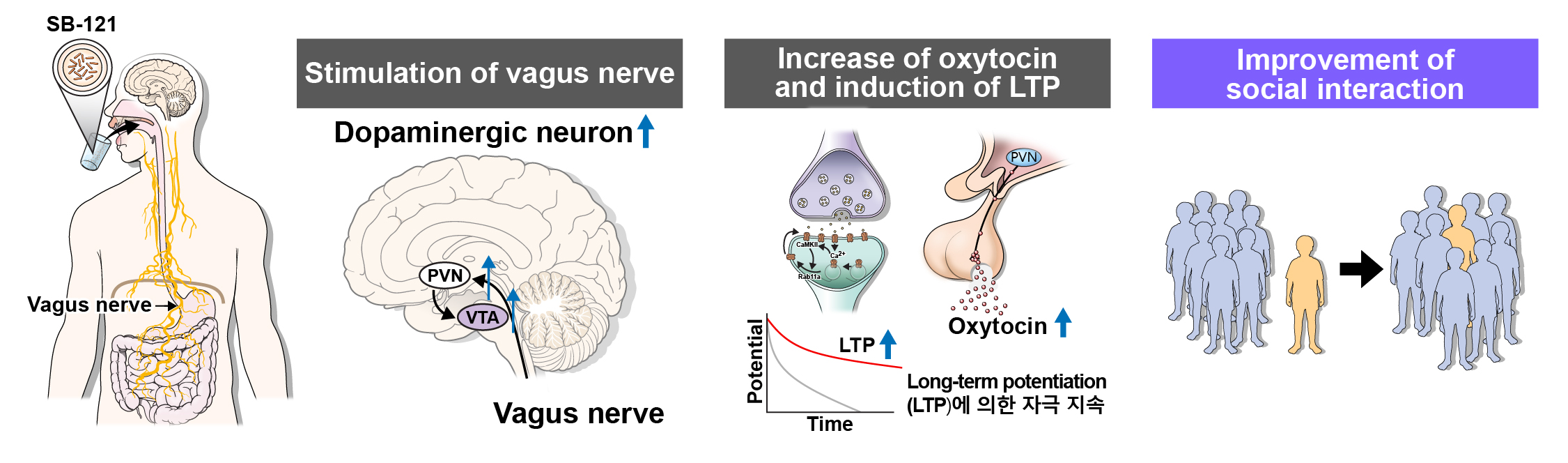
‘SB-121’ is a clinical-stage pipeline under development by Scioto Biosciences, a US biotech acquired by Genome & Company in August 2020.
‘SB-121’ is a lead pipeline of Scioto Biosciences, Genome & Company's subsidiary, which targets CNS (gut-brain axis) disorders. It is developed by using Lactobacillus.reuteri (L.reuteri) strain isolated from breast milk of a healthy mother. A proprietary drug formulation platform technology (ABT, Activated Bacterial Therapeutics) developed by Scioto Biosciences was applied.
Many studies have confirmed the effects of Lactobacillus.reuteri strain that activates the secretion of oxytocin which then affects the relieving of symptoms of autism spectrum disorder (ASD). Oxytocin upregulation and anti-inflammatory effects in gut have been confirmed in pre-clinical studies.
‘SB-121’ is being developed for treatment of gut-brain axis related disorders, such as autism and gut injury related diseases, namely necrotizing enterocolitis (NEC) and clostridium difficile infection (CDI).
Through Phase 1 clinical study safety and efficacy has been verified. Subsequently, Phase 2 study will be conducted targeting both autism and also necrotizing enterocolitis patients.
*Find more details at Scioto Biosciences website.

GEN-004Obstetrics disease microbiome treatment

‘GEN-004’ is a candidate for microbiome therapeutic that has shown an in-vivo efficacy of increasing implantation of embryo.
‘GEN-004’ is being developed as an ovule formulation of live biotherapeutic products(LBP) single bacterial strain that is isolated from healthy human.
‘GEN-004’ inhibits immune responses that hinder embryo implantation and has antibacterial effects against harmful bacteria that cause vaginitis. The efficacy for increasing the embryo implantation rate with ‘GEN-004’ has been confirmed in in-vivo infertility model with induction of inflammation. 'GEN-004' is currently in pre-clinical stage.
GEN-501Skin diseases microbiome treatment

'GEN-501' is a candidate for microbiome therapeutic that is under development by using Cutibacterium avidum (C.avidum) single strain that is isolated from the skin of healthy human.
‘GEN-501’ is being developed in the formulation for local application to the skin such as ointment. 'GEN-501' selectively inhibits Staphylococcus aureus (S.aureus) which is known to be the main cause of atopic dermatitis and cancer associated rash. 'GEN-501' is currently in preclinical study and is expected to enter phase 1 clinical study in 2022. 'GEN-501' is also expected to expand the target indications to other skin diseases occurred by the abundance of 'S. aureus' based on the progress of the clinical development.
Debio 0633
In May 2024, Genome & Company out-licensed an antibody for novel target ADC application to the global pharmaceutical company Debiopharm. ‘Debio 0633’ is an ADC (Antibody Drug Conjugate) candidate being developed by combining the antibody for novel target ADC application developed by Genome & Company with Debiopharm’s Multilink™ technology.
GENA-104 Novel-target ICI
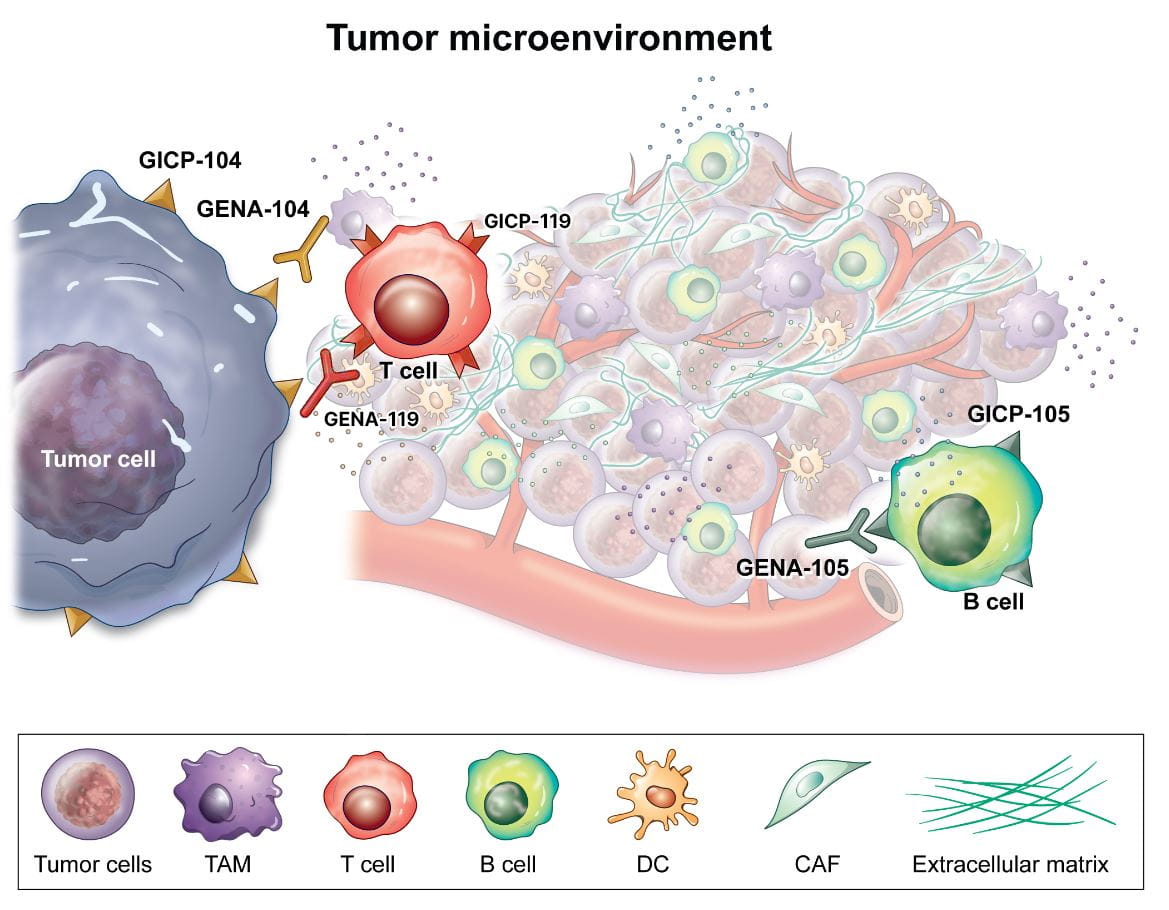
Genome & Company specializes in discovery and validation of novel targets for anti-cancer applications.
Novel targets identified by Genome & Company strongly inhibits immune cells (T cells) than PD-L1, the best known immune checkpoint. Once 'GEN-104' is developed into novel target immuno-oncology therapeutic, it is expected to show enhanced activation levels of immune cells (T-cells) than existing immune checkpoint inhibitors. It is our vision to provide new treatment options to patients who are non-responsive or resistant to existing immunotherapy.
Our leading novel target 'GEN-104' is a 'first-in-class' immune checkpoint inhibitor discovered by Genome & Company. It inhibits novel target (CNTN4) that suppresses T cell responses on tumor cells. Pre-clinical research has confirmed its anti-cancer effect, and the mechanism of action has been presented at American Association of Cancer Research (AACR) in 2021~2024.
GENA-105 Novel-target ICI

Genome & Company specializes in discovery and validation of novel targets for anti-cancer applications.
‘GENA-105’ is a new antibody drug that induces the activation of immune cells (T-cells) and anticancer effects by surpressing the novel target (TLT2) expressed in B cells. The study results on immune cell (T cell) suppression effect of the novel target TLT2 were presented in American Association for Cancer Research (AACR) 2022.
Currently, the final candidate for ‘GENA-105’ has been chosen and confirmed its safety in NHP (non-human primate) settings, and the IND enabling study is under progress.
GENA-119 Novel-target ICI

‘GENA-119’ is a novel immune checkpoint inhibitor candidate targeting the APP, a newly discovered immune checkpoint protein by Genome & Company.
'GENA-119' is a novel immune checkpoint inhibitor candidate targeting the APP, a newly discovered immune checkpoint protein by Genome & Company.
APP is a binding receptor on the T cell surface that is expressed by cancer cells. It has been observed that APP inhibits the proliferation and activation of T cells. Therefore, 'GENA-119' is expected to enhance the anti-tumor efficacy of T cells by suppressing the immune-inhibitory activity of APP expressed on T cells and inducing their activation.
GENA-111 ADC
‘GENA-111’ is an ADC (Antibody-Drug Conjugate) candidate, which is a novel target CD239 antibody-based ADC therapeutic substance.
Genome & Company has entered into a partnership with the global pharmaceutical company DiBiopharm in 2021 to collaborate on joint research and development.
Leveraging Genome & Company's drug development platform called GNOCLE™, we are discovering multiple antibodies against new tumor targets and conducting research and development of ADC candidates. DiBiopharm's Multilink™ technology is being incorporated to precisely deliver the antibodies to the antigens.

