Genome & Company
Contact
지놈앤컴퍼니
경기도 수원시 영통구 창룡대로256번길 50, 광교플렉스데시앙 7층 (우 16229)
+82-31-628-0150 / +82-70-8673-2440
개인정보의 수집 및 이용동의 [필수]
주식회사 지놈앤컴퍼니는 (이하 '회사'는) 고객님의 개인정보를 중요시하며, “개인정보 보호법”, "정보통신망 이용촉진 및 정보보호" 등에 관한 법률을 준수하고 있습니다. 회사는 개인정보취급방침을 통하여 고객님께서 제공하시는 개인정보가 어떠한 용도와 방식으로 이용되고 있으며, 개인정보보호를 위해 어떠한 조치가 취해지고 있는지 알려드립니다. 회사는 개인정보취급방침을 개정하는 경우 웹사이트 공지사항(또는 개별공지)을 통하여 공지할 것입니다.
1. 처리하는 개인정보 항목 및 수집방법
회사는 Contact (일반문의, 사업개발문의, IR문의, 미디어문의 등) 서비스 운영을 위해 아래와 같은 개인정보를 수집하고 있습니다.
- 필수정보: 이름, 이메일, 연락처, 소속단체, 문의내용
- 수집방법: 회사는 홈페이지를 통한 문의 사항에 대해 홈페이지 내 Contact 게시판 페이지를 통해 개인정보를 수집 및 이용하고 있습니다.
‘필수 정보 수집’ 동의를 거부할 권리가 있으며, 필수 정보 외 선택 정보의 수집에 동의를 거부할 수 있습니다. 필수 정보 수집의 동의를 하지 않을 경우 Contact 서비스 이용이 제한될 수 있습니다.
서비스 이용과정에서 아래와 같은 정보들이 자동으로 생성되어 수집될 수 있습니다.
- IP Address, 쿠키, 방문 일시, 서비스 이용 기록
2. 개인정보 처리 목적
회사는 수집한 개인정보를 다음의 목적을 위해 활용합니다.
- Contact 서비스 이용에 따른 본인확인, 개인 식별, 문의사항 전달 및 결과 고지 등
3. 개인정보의 처리 및 보유기간
원칙적으로, 개인정보 수집 및 이용목적이 달성된 후에는 해당 정보를 지체 없이 파기합니다. 단, 관계법령의 규정에 의하여 보존할 필요가 있는 경우 회사는 일정한 기간 동안 회원정보를 보관할 수 있습니다.
- 보존 항목 : 문의접수시 기재한 개인정보 및 상담내용
- 보존 근거 : 전자상거래 등에서의 소비자보호에 관한 법률(소비자의 불만 또는 분쟁처리에 관한 기록)
- 보존 기간 : 3년
4. 개인정보의 파기에 관한 사항
회사는 원칙적으로 개인정보 수집 및 이용목적이 달성된 후에는 해당 정보를 지체없이 파기합니다. 파기절차 및 방법은 다음과 같습니다.
1) 파기절차
입력하신 정보는 목적이 달성된 후 별도의 데이터베이스(database)로 옮겨져(종이의 경우 별도의 서류함) 내부 방침 및 기타 관련 법령에 의한 정보보호 사유에 따라(보유 및 이용기간 참조) 일정 기간 저장된 후 파기되어집니다.
별도 데이터베이스로 옮겨진 개인정보는 법률에 의한 경우가 아니면 보유되어지는 이외의 다른 목적으로 이용되지 않습니다.
2) 파기방법
전자적 파일형태로 저장된 개인정보는 기록을 재생할 수 없는 기술적 방법을 사용하여 삭제합니다. 종이에 출력된 개인정보는 분쇄기로 분쇄하거나 소각을 통하여 파기하고 있습니다.
5. 개인정보처리의 위탁에 관한 사항
회사는 제3자에게 개인정보를 위탁하고 있지 않으며, 추후 위탁계약이 발생할 경우, 홈페이지를 통해 고지하고, 본 ‘개인정보처리방침’의 내용을 갱신하도록 하겠습니다.
6. 이용자 및 법정대리인의 권리 및 그 행사방법에 관한 사항
이용자 및 법정 대리인은 언제든지 등록되어 있는 자신 혹은 당해 만 14세 미만 아동의 개인정보를 조회하거나 수정할 수 있으며 삭제를 요청할 수도 있습니다. 개인정보보호 책임자에게 서면, 전화 또는 이메일로 연락하시면 본인 확인 절차를 거친 후 지체 없이 조치하겠습니다.
이용자가 개인정보의 오류에 대한 정정을 요청한 경우에는 정정을 완료하기 전까지 당해 개인정보를 이용 또는 제공하지 않습니다. 또한 잘못된 개인정보를 제3자에게 이미 제공한 경우에는 정정 처리결과를 제3자에게 지체 없이 통지하여 정정이 이루어지도록 하겠습니다.
회사는 이용자 혹은 법정 대리인의 요청에 의해 해지 또는 삭제된 개인정보는 ‘3. 개인정보의 처리 및 보유기간’ 및 ‘4. 개인정보의 파기에 관한 사항’에 명시된 바에 따라 처리, 파기하고 그 외의 용도로 열람 또는 이용할 수 없도록 처리하고 있습니다.
7. 개인정보보호 책임자 및 담당 부서 등 안내
회사는 고객의 개인정보를 보호하고 개인정보와 관련한 문의를 처리하기 위하여 아래와 같이 관련 부서 및 개인정보관리책임자를 지정하고 있습니다.
개인정보보호 책임자
성 명: 서영진
소속부서: 경영관리부문
지위: 부사장
전화번호: 031-620-0150
Email: youngjin.seo@genomecom.co.kr
개인정보보호 담당자
성 명: 설수기
소속부서: PR팀
지위: 팀장
전화번호: 070-4821-3840
Email: sugi@genomecom.co.kr
귀하께서는 회사의 서비스를 이용하시며 발생하는 모든 개인정보보호 관련 민원을 개인정보책임자 혹은 담당부서로 접수하실 수 있습니다. 회사는 이용자들의 문의사항에 대해 신속하게 충분한 답변을 드릴 것입니다.
기타 개인정보침해에 대한 신고나 상담이 필요하신 경우에는 아래 기관에 문의하시기 바랍니다.
- 개인정보침해신고센터 (privacy.kisa.or.kr / 국번없이 118)
- 개인정보분쟁조정위원회 (kopico.go.kr / 1833-6972)
- 대검찰청 사이버수사과 (www.spo.go.kr / 국번없이 1301)
- 경찰청 사이버안전국 (http://cyberbureau.police.go.kr / 국번없이 182)
8. 고지의 의무
현 개인정보처리방침 내용 추가, 삭제 및 수정이 있을 시에는 홈페이지의 ‘공지사항’을 통해 사전 고지할 것입니다.
9. 기타
회사 홈페이지에 링크 되어있는 웹사이트들이 개인정보를 수집하는 행위에 대해서는 본 ‘개인정보처리방침’이 적용되지 않음을 알려드립니다.



Pipeline
GNOCLE™ 플랫폼
지놈앤컴퍼니는 '임상데이터기반 연구개발(Bed-to-Bench)" 전략을 토대로 신약 발굴 플랫폼 지노클(GNOCLE™)을 구축하여 마이크로바이옴 치료제 및 신규타깃 면역항암제를 연구개발 하고 있습니다. 연구중심병원의 풍부한 임상데이터를 바탕으로 신약개발 프로세스에서의 실패위험을 낮춤으로써, 혁신 마이크로바이옴 및 신규타깃기반 의약품 후보물질을 효과적으로 연구개발하고 있습니다.
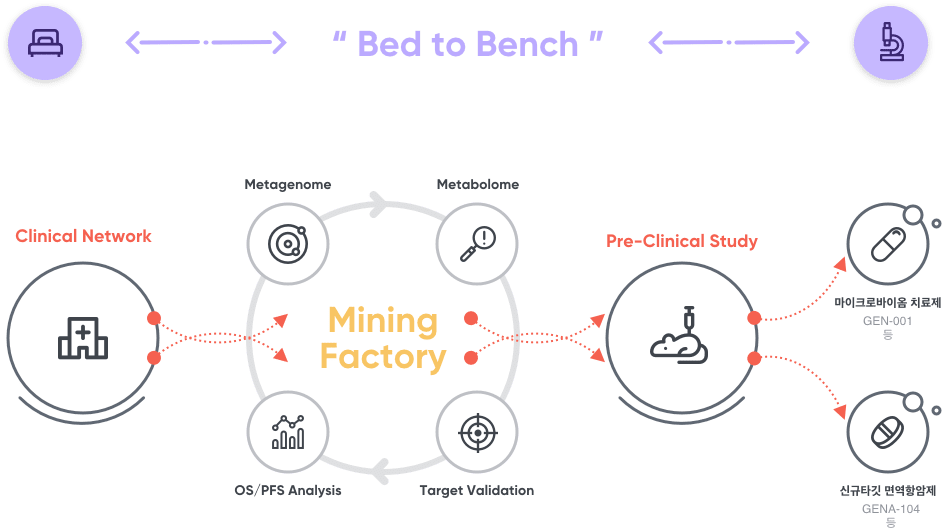
- 01 임상 데이터베이스 구축
- [마이크로바이옴] 기존 의약품으로 치료효과가 낮은 질환들의 환자 마이크로바이옴 샘플 확보 (암, 항암발진, 아토피, 치매, 난임, 인지기능 등 핵심질환 관련 10,000여개 임상샘플 보유)
- [신규타깃] 항암치료를 받은 암환자들의 암조직분석을 통해 기존치료제 불응성 환자들에 적용 가능한 신규타깃 라이브러리 구축
- 02 마이크로바이옴 후보물질 도출 및 신규 약물표적 검증
- [마이크로바이옴] 확보된 임상샘플을 기반으로, 다중체학(Multi-Omics) 연구를 통한 작용기전규명 및 효능검증 시험을 통해 마이크로바이옴 의약품 후보물질 도출
- [신규타깃] 구축된 신규타깃 라이브러리를 대상으로 해당 타깃 발현여부에 따른 환자 예후비교 및 타깃의 항암효능/작용기전 등을 규명하여 신규타깃 검증 (이후 검증된 신규타깃에 기반한 신규 면역항암제 후보물질을 도출과정을 거침)
신약 연구소 Pipelines
* 표 안의 파이프라인명을 클릭하시면 자세한 내용을 확인할 수 있습니다.
| Modality | Pipeline | Tartget | Indication | Developmental Status | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Target | Hit | Lead | Nonclinical candidate | IND enabling | Phase I | Partner | ||||
|
ADC
|
Debio 0633
|
N/D | Solid tumors | License-Out(May 2024) |
 |
|||||
|
GENA-104
|
CNTN4 | Solid tumors | ||||||||
| GENA-120 | ITGB4 | Solid tumors | ||||||||
| GENB-120 | ITGB4 TROP2 |
Solid tumors | ||||||||
| GENA-121 | N/D | Solid tumors | ||||||||
| ADC Programs | N/D | N/D | ||||||||
|
mAb(Immuno-oncology)
|
EP0089 | CNTN4 | Solid tumors |
KDDF Funded IND Approval (MFDS)
|
License-Out(Feb 2025) |
 |
||||
|
GENA-119
|
APP | Solid tumors |
KDDF Funded
|
|||||||
|
NCE
|
GENC-116 | NUAK1 | Fibrosis |
KDDF Funded
|
||||||
* N/D, not disclosed; mAb, monoclonal antibody; ADC, antibody-drug conjugate; Nce, new chmical entity
* License-Out 완료된 파이프라인은 파트너사가 연구개발을 담당하고 있어 실제 연구개발 진행 사항과 다소 차이가 발생할 수 있습니다.
GEN-001 면역항암 마이크로바이옴 치료제
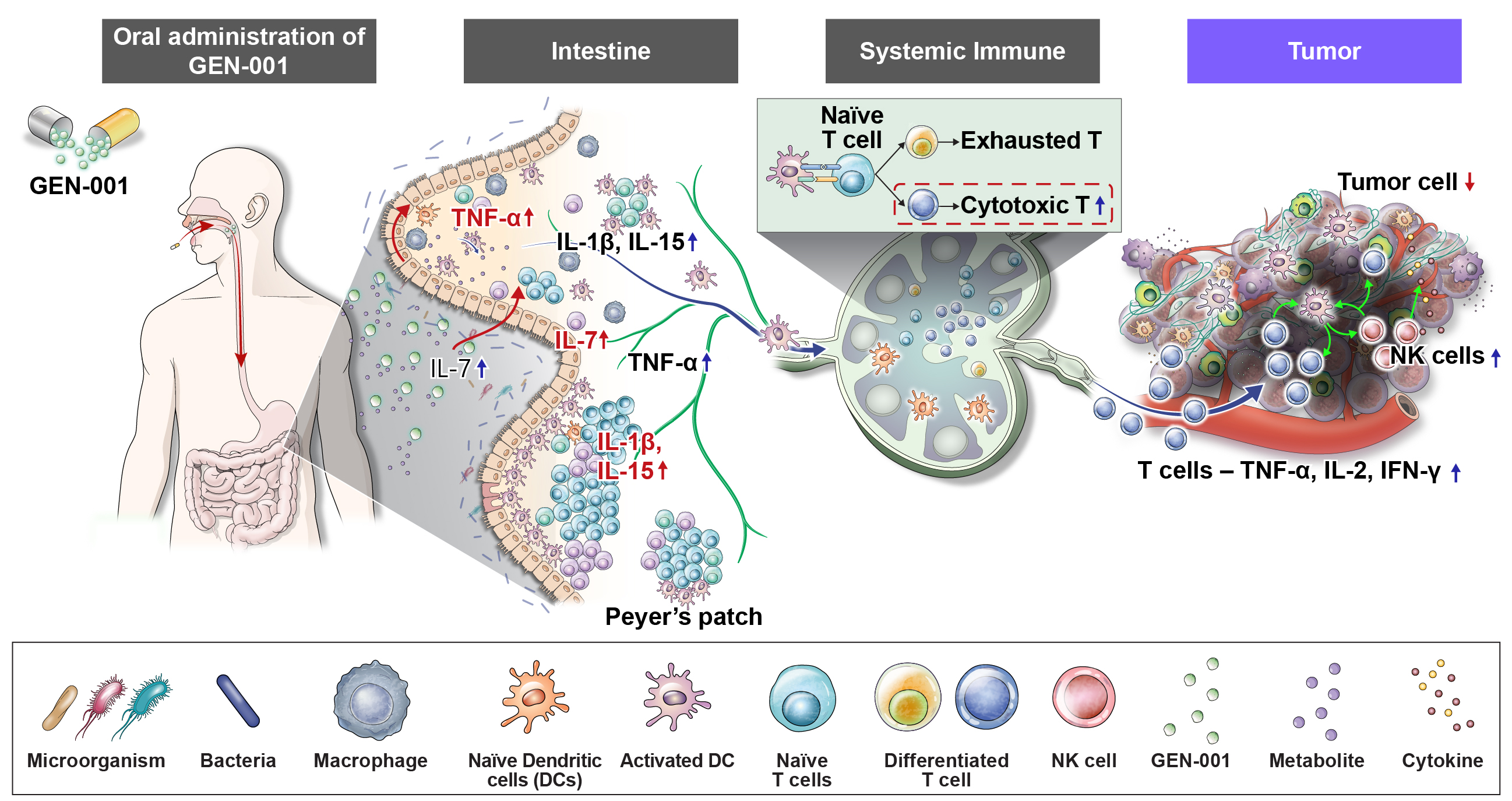
지놈앤컴퍼니의 주력 파이프라인 'GEN-001'은 국내 최초로 개발 중인 면역항암 마이크로바이옴 치료제 후보물질입니다.
'GEN-001'은 건강한 사람에서 분리 동정한 락토코커스 락티스(Lactococcus lactis, 이하 L. lactis) 단일균주(single strain)를 주성분으로 한 경구용 마이크로바이옴 치료제 후보물질입니다. 암환자의 면역력 활성화를 통한 면역항암 효능을 가지고 있습니다. 전임상 연구에서 'GEN-001'은 단독 면역항암효능 뿐 아니라, 면역항암제(면역관문억제제)와의 병용 면역항암효능을 확인한 바 있습니다. 현재 'GEN-001'은 독일 머크 바벤시오®와 위암을 대상으로 병용 임상 2상시험을 진행 중입니다. 또한 MSD(미국 머크)의 키트루다®와 담도암 대상 병용 임상 2상시험을 진행 중에 있습니다.
- [파트너십]
- Merck KGaA 공동 연구개발: 'GEN-001'과 면역관문억제제 (Bavencio®) 병용임상시험 진행 중
- MSD 공동 연구개발: 'GEN-001'과 면역관문억제제 (KEYTRUDA®) 병용임상시험 진행 중
- [사업화성과]
- LG화학 동아시아 지역 기술이전 체결
SB-121뇌질환 마이크로바이옴 치료제
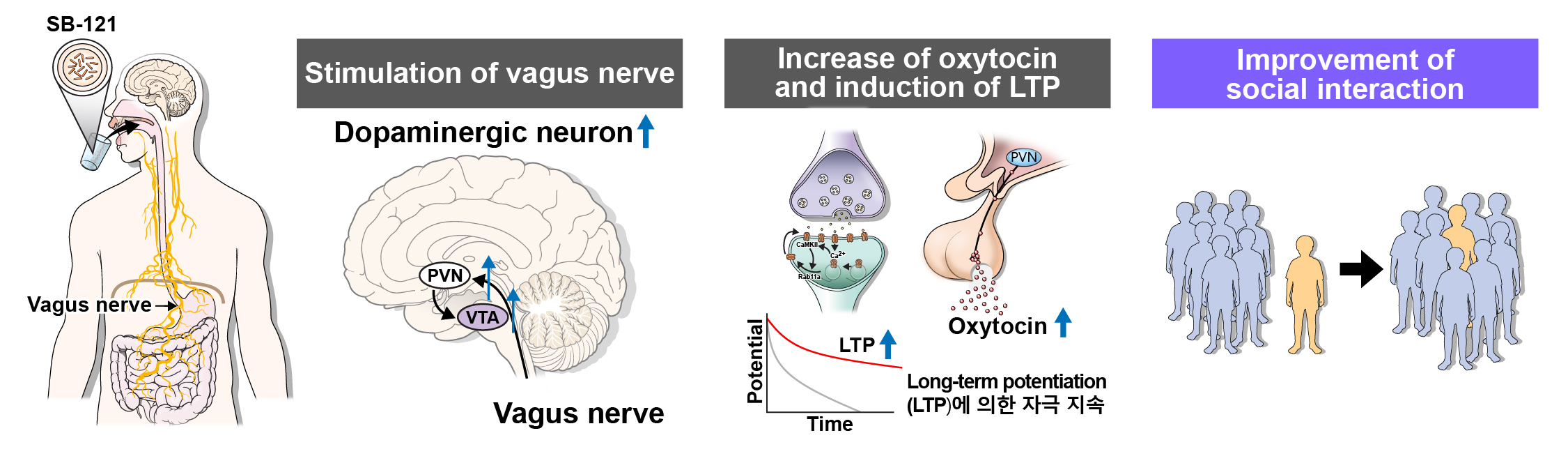
‘SB-121’은 2020년 8월 지놈앤컴퍼니가 인수한 미국 바이오텍 Scioto Biosciences의 핵심 파이프라인입니다.
‘SB-121’은 건강한 산모의 모유로부터 유래된 락토바실러스 루테리(Lactobacillus.reuteri 이하 L.reuteri) 균주를 주성분으로 한 당사의 자회사 Scioto Biosciences가 보유한 뇌질환 치료제입니다. Scioto Biosciences가 보유한 특수제형플랫폼기술(ABT platform, Activated Bacterial Therapeutics)이 적용되었습니다. L. ruteri 균주는 다수 임상연구를 통해 자폐증 완화에 영향을 미치는 옥시토신 분비를 활성화하는 효과가 있음을 확인된 효능균주입니다. 전임상 연구에서 ‘SB-121’은 장내 상처 완화와 옥시토신 분비를 활성화하는 효과가 있음이 확인됐습니다. 자폐증 대상 임상 1상을 통해 안전성과 내약성이 확인됐으며, 자폐증 뿐만 아니라 신생아괴사성장염까지 확대하여 임상2상을 진행할 예정입니다.
* 자세한 사항은 Scioto Biosciences에서 확인가능합니다.

GEN-004산과질환 마이크로바이옴 치료제
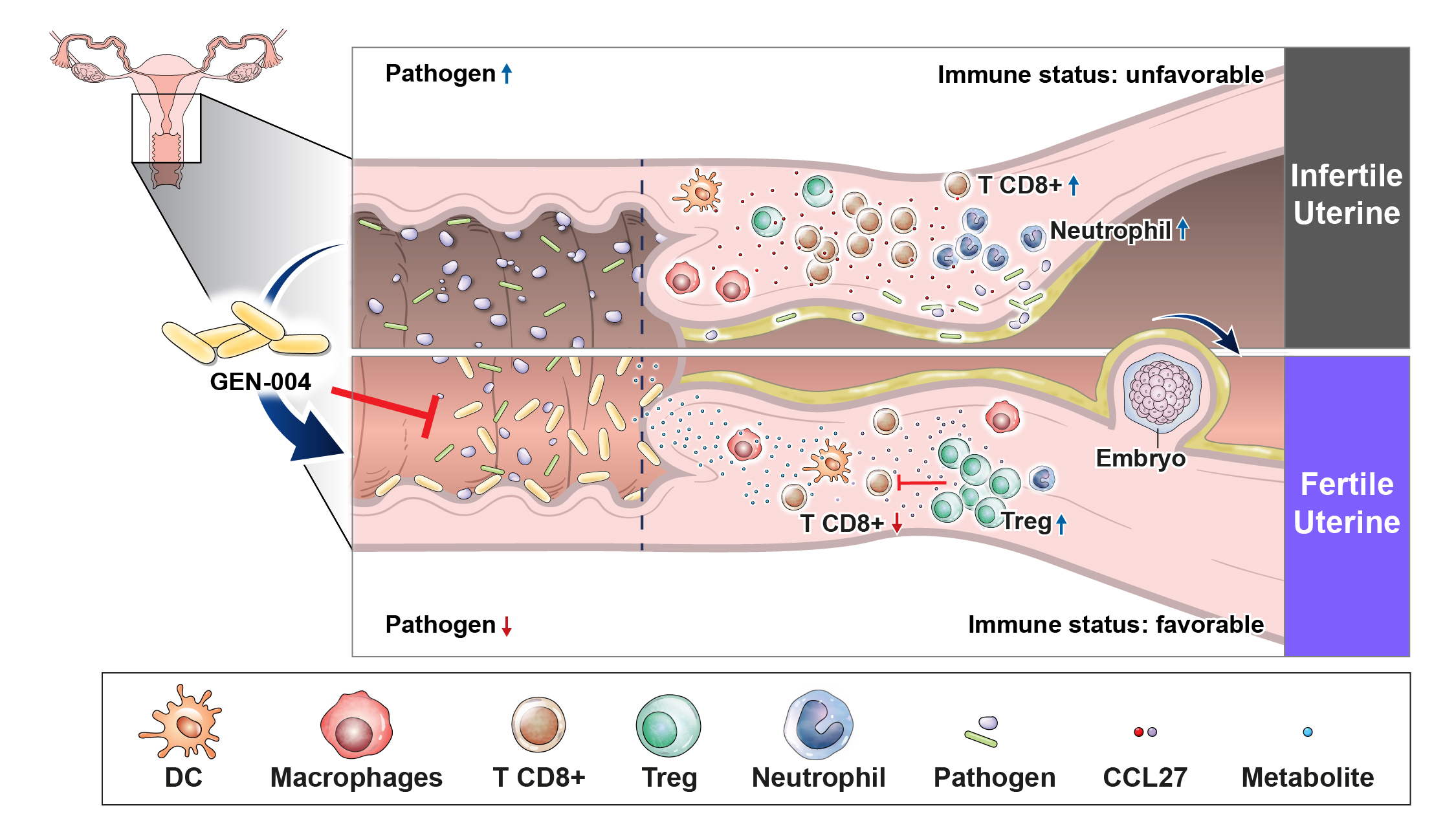
‘GEN-004’는 국내 최초로 개발 중인 시험관 아기 시술의 착상 증진 효능을 가진 마이크로바이옴 치료제 후보물질입니다.
‘GEN-004’는 건강한 사람에서 분리 동정한 단일균주(Single strain)를 주성분으로 한 질좌제형으로 개발 중입니다. ‘GEN-004’는 배아착상을 방해하는 면역반응 억제 및 질염유발 유해균에 대한 항균 효능을 가지고 있는데, 구체적으로 염증 유발 난임실험모델에서 ‘GEN-004’로 인한 배아 착상률 증진효능이 확인된 바 있습니다. ‘GEN-004’는 현재 전임상 진행 중입니다.
GEN-501피부질환 마이크로바이옴 치료제
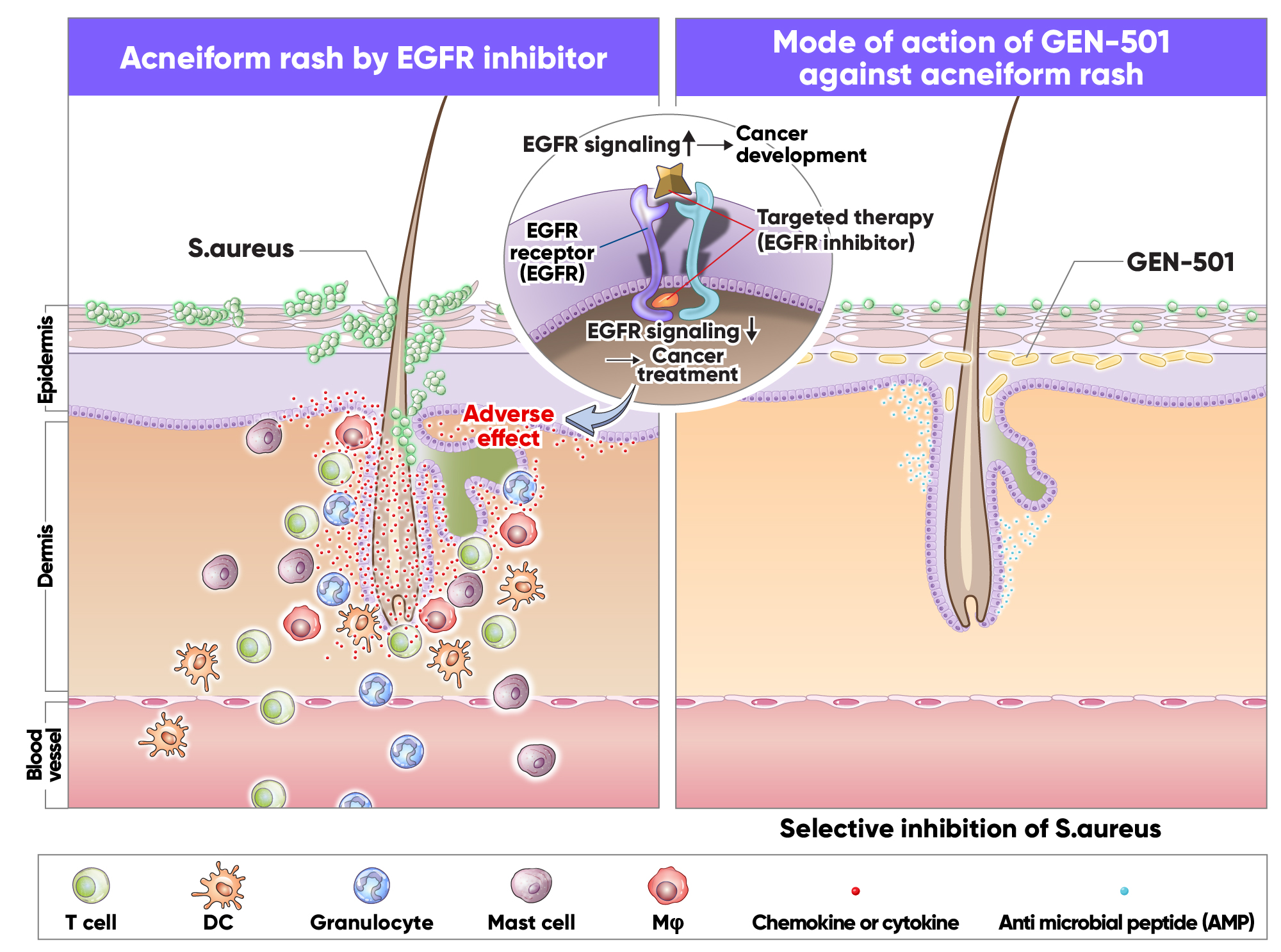
'GEN-501'은 건강한 사람의 피부에서 분리 동정한 피부상재 큐티박테리움 아비덤(Cutibacterium avidum 이하 C.avidum) 단일균주(single strain)를 주성분으로 개발되고 있는 마이크로바이옴 치료제 후보물질입니다.
'GEN-501'은 피부에 바르는 도포제형으로 개발되고 있으며, 아토피 피부염과 항암발진 등의 원인균으로 알려진 황색포도상구균(Staphylococcus aureus 이하 S.aureus)을 선택적으로 억제하는 효능을 갖고 있습니다. 'GEN-501'은 현재 전임상 연구 중입니다. 향후 초기 임상시험결과를 기반으로 'GEN-501'은 'S. aureus' 우점양성으로 인해 발생하는 다른 피부 질환으로 적응증을 확장할 계획입니다.
Debio 0633
2024년 5월 지놈앤컴퍼니는 글로벌 제약회사 디바이오팜(Debiopharm)에 신규타깃 ADC용 항체를 기술이전하였습니다. 'Debio 0633'은 지놈앤컴퍼니가 개발한 신규타깃 ADC용 항체와 디바이오팜의 Multilink™(멀티링크) 기술을 결합하여 개발 중인 ADC(Antibody Drug Conjugate) 치료제 후보 물질입니다.
GENA-104 신규타깃 면역항암제
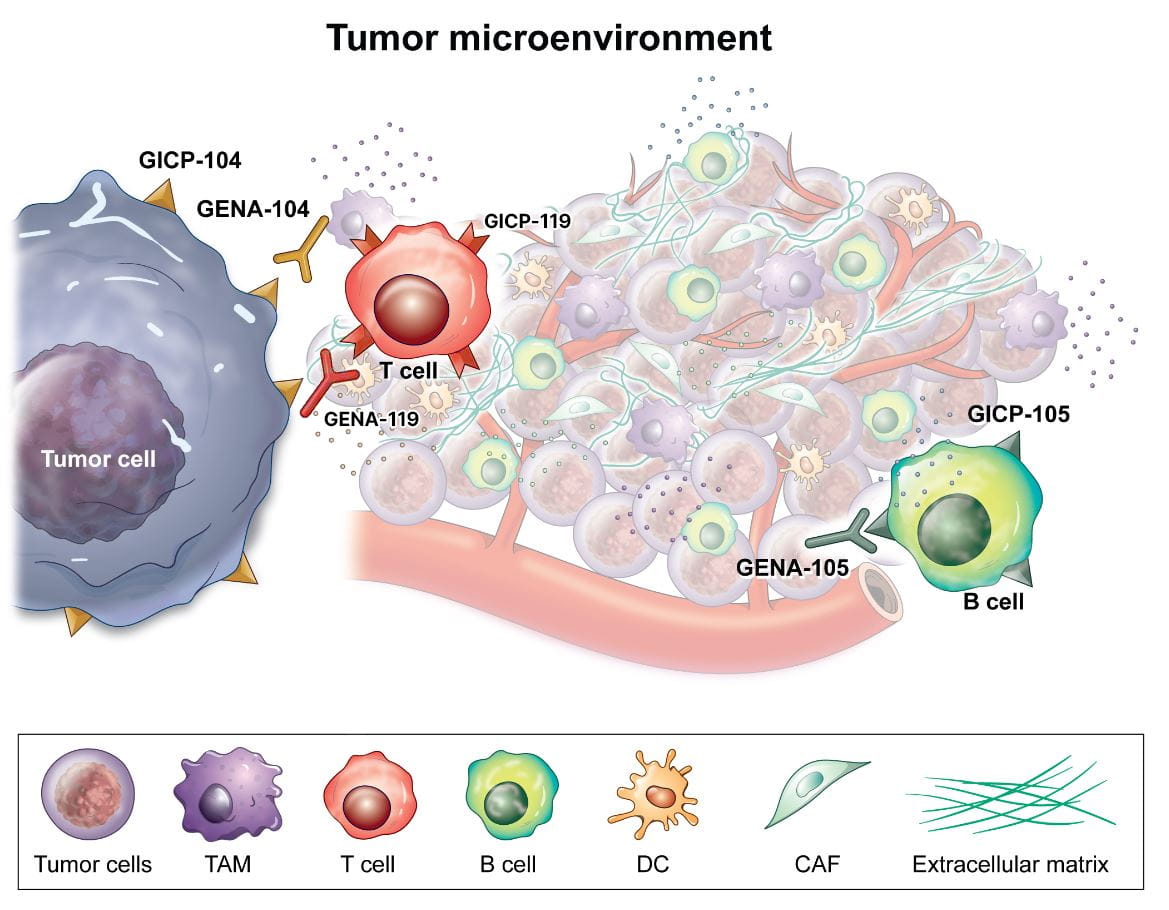
지놈앤컴퍼니는 신규타깃(Novel target)을 자체 발굴하고, 이렇게 발굴된 신규타깃을 타깃으로 하는 신약을 개발하고 있습니다.
지놈앤컴퍼니가 발굴한 신규타깃은 대표적 면역항암 타깃인 PD-L1보다 면역세포(T세포)를 강하게 억제하는 특성을 보이고 있습니다. 해당 신규타깃을 억제하는 면역항암제를 개발할 경우 기존 면역항암제 대비 우수한 면역세포(T세포) 활성효과가 기대됩니다. 당사는 이러한 신규타깃 면역항암제 개발을 통해 기존 면역항암제 비반응 환자군에게 새로운 치료제를 제공하고자 합니다.
‘GENA-104’는 자체발굴한 신규타깃 CNTN4를 억제하는 항체로, 전임상 연구를 통해 ‘GENA-104’가 면역세포(T세포)를 활성화시켜 효과적으로 암세포를 사멸하는 효능이 확인되었습니다. 이러한 결과는 2021년 부터 2024년까지 4년 연속 미국암연구학회(AACR)에서 공식 발표된 바 있습니다. 또한 신규타깃 CNTN4가 암세포에서만 특이적으로 과발현되는 특성을 활용하여 ‘GENA-104’를 신규타깃 ADC용 항체로도 연구개발 중입니다.
GENA-119 신규타깃 면역항암제

지놈앤컴퍼니는 신규타깃(Novel target)을 자체 발굴하고, 이렇게 발굴된 신규타깃을 목표로 하는 면역항암제를 개발하고 있습니다.
'GENA-119'는 신규 면역항암타겟인 APP를 표적으로 하는 면역관문억제제 후보물질입니다. APP는 지놈앤컴퍼니가 발굴한 신규 면역관문단백질입니다.
APP는 암세포에서 발현하는 CNTN-4의 T 세포 표면의 결합 수용체로 T 세포의 증식과 활성을 억제하는 것을 확인하였습니다.
이에, 'GENA-119'는 T 세포에서 발현하는 APP의 면역억제활성을 억제하여 T 세포의 활성을 유도함으로써 T 세포의 항 종양 효능을 증가시킬 것으로 기대하고 있습니다.
GENA-111-ADC
'GENA-111'은 ADC 후보 물질로, 신규 타깃 CD239 항체 기반 ADC 치료 물질입니다.
2021년 글로벌 제약회사 디바이오팜과 파트너십을 맺고 공동연구개발을 진행하고 있습니다.
지놈앤컴퍼니의 신약 개발플랫폼 지노클(GNOCLE™)을 기반으로 새로운 종양 표적에 대한 여러 항체를 발굴하고 항체를 항원에 정확하게 전달하는 디바이오팜의 ‘멀티링크(Multilink™)’기술을 접목해 ADC 후보 물질 연구개발을 하고 있습니다.